Share
Amyotrophic Lateral Sclerosis (ALS) presents one of the most formidable challenges in medical science. For too long, the search for effective treatments has been hampered by a temptation to revisit familiar hypotheses and established biological pathways. While understandable given the slow progress and frequent setbacks, this approach rarely advances the science.
At the heart of insitro’s collaboration with Bristol Myers Squibb (BMS) is the deliberate decision to boldly seek novel biology. We generated billions of multimodal data points from human-derived cellular systems and used machine learning and AI to guide our understanding of ALS biology. Our unbiased approach revealed novel biological targets – points of intervention that have been previously invisible.
Today, we are excited to announce an expansion of our collaboration with BMS toward the development of new therapeutic approaches for the treatment of ALS. BMS nominated two additional ALS targets – ALS-2 and ALS-3 – under our collaboration.
insitro will advance its own oligonucleotide program for ALS-1 while simultaneously progressing a small molecule for BMS. This expansion underscores the value of taking a fundamentally different approach in looking for treatments in a disease that has challenged every conventional approach.
The Virtual HumanTM: Letting the Data Lead
ALS is heterogeneous, its biology is complex, and many of the most intuitive hypotheses have already been tested, mostly unsuccessfully. This is precisely where insitro’s Virtual Human™ Causal Discovery Platform becomes uniquely enabling. Our platform is built on the premise that to truly understand the drivers of complex disease, one must measure it across multiple biological domains and modalities. This allows us to employ machine learning to disentangle intricate biology and uncover patterns that are invisible to conventional human inspection.
To demonstrate the power of this premise, we worked with over 200 distinct ALS cell lines – both engineered models and patient-derived lines, spanning familial (inherited) and sporadic disease. We developed protocols to generate motor neurons robustly and rapidly enough to support large-scale experimentation while maintaining disease-relevant biology. Crucially, we established an imaging and screening infrastructure, including our POSH technology for pooled optical screening, capable of measuring thousands of cellular features across millions of perturbed cells. This generates the sheer data density required to power AI-driven causal inference.
By integrating massive-scale human-derived cellular data with AI, our Virtual Human allows us to identify causal disease processes using an unbiased, data-driven approach. We were able to discover ALS signatures using iPSC-derived motor neurons and used those signatures to pinpoint novel therapeutic targets – molecular intervention points capable of modulating the effects of TDP-43 dysfunction, a core disease mechanism implicated in nearly 97% of ALS patients. We validated that modulation of these targets reverts key pathologies caused by a loss of TDP-43 protein, leading to rescue of neurite growth in cellular models of ALS – a meaningful milestone that reflects structural repair in human neurons.
From Hidden Signatures to Master Regulators
TDP-43 pathology, where the protein leaves the nucleus and aggregates in the cytoplasm, is documented in 97% of ALS patients. While the field has known of this pathology for years, knowing it exists is fundamentally different from knowing precisely how to stop this from occurring.
A pivotal insight emerged from observing models derived from genetically distinct ALS mutations (C9orf72, VCP, and TDP‑43) that displayed convergent morphological phenotypes correlating with measured TDP-43 pathology. insitro’s machine learning models, trained on millions of high-dimensional cellular images, resolved what conventional inspection could not: a shared latent pattern of dysfunction underlying these variants. This unified signature illuminated a common downstream pathway that now guides our therapeutic efforts.
Moving from this shared biology to a robust therapeutic standard was the next critical step. Our BMS colleagues, drawing on decades of neuroscience expertise, pushed us to look beyond simple protein markers to focus on truly meaningful outcomes. TDP-43 regulates proteins essential for neuronal health, and when it misbehaves in ALS, these critical proteins decrease due to improper splicing.
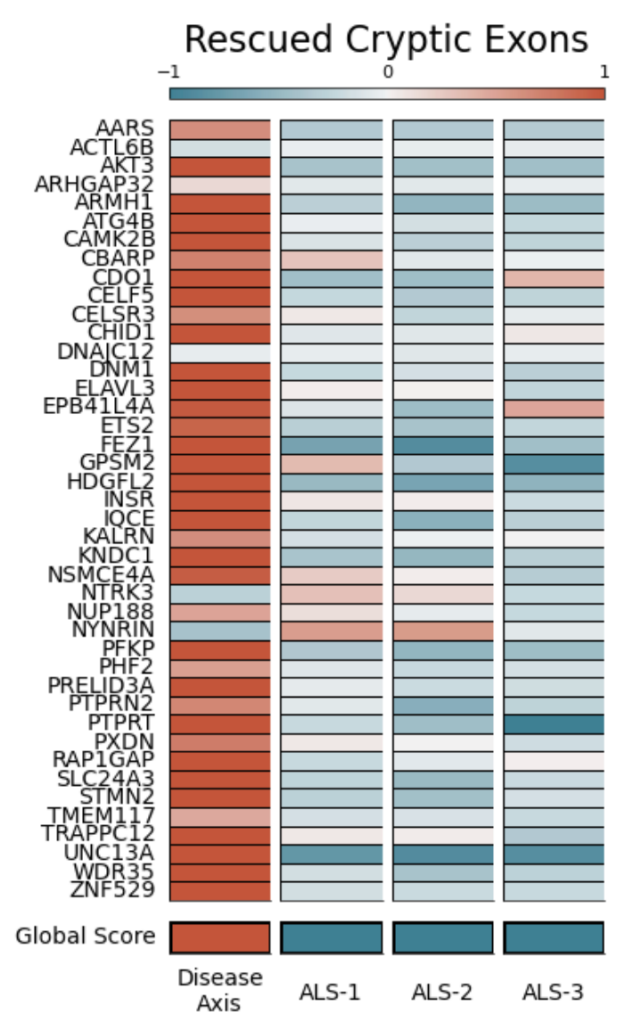
ALS-1, ALS-2 and ALS-3 knockdown rescues splicing defects across key TDP-43 targets and restores proteins affected by TDP-43 h
Recent clinical tests on patient tissue and CSF confirm these same loss-of-splicing signatures, validating that this is a disease-relevant mechanism. By optimizing our high-dimensional AI-enabled platform to track panels of “cryptic exon” disease signatures, we are able to identify targets that revert the ensemble of both ML-derived and molecular disease signatures, enriching ones with maximum patient impact.
Furthermore, we converged on neurite outgrowth – the actual structural repair of a human motor neuron – as a critical validation experiment. This decision raised the bar considerably, but we demonstrated that modulation of our targets leads to rescue of neurite growth. This means the targets we advanced are not merely modulating surrogate biomarkers; they are demonstrating the capacity to restore fundamental neuronal architecture and function.
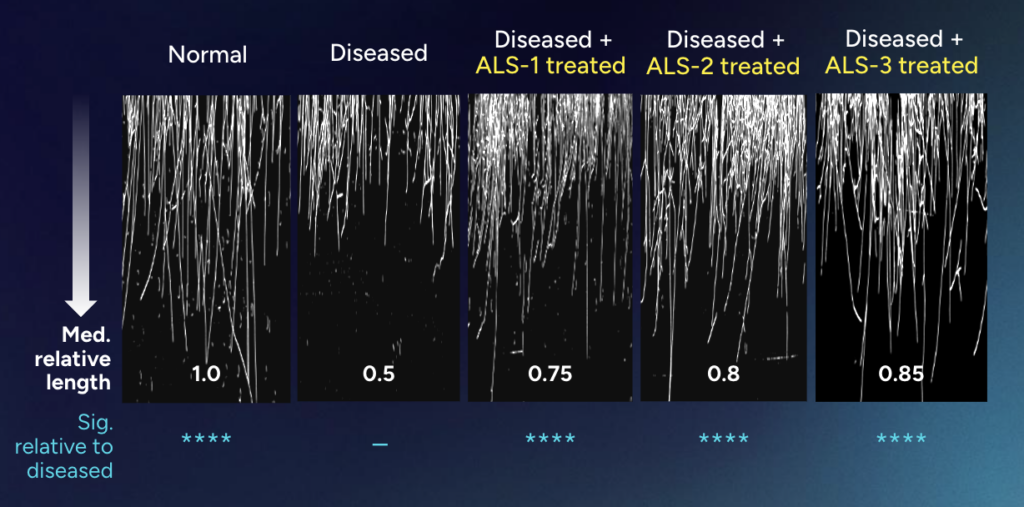
Targeting this discovered pathway rescues neurite regrowth defect in ALS diseased human motor neurons
Rescuing human motor neuron regrowth: The lost connection of muscle to neurons is a hallmark of ALS causing paralysis. To test insitro’s CellML predictions on a relevant injury model to a patient setting, in a cellular model of axotomy where neuron axons are cut while under diseased conditions to observe regrowth capacity.
Our results suggest that our targets are not making incremental changes in a single disease marker. Rather, they are master regulators: nodes in the causal network that, when perturbed, can reverse core pathological features of the disease. This neurite rescue phenotype suggests we have found intervention points capable of addressing a biological convergence point for multiple ALS-causing familial mutations – which together only cover 10% of ALS patients – as well as the 90% of patients with sporadic ALS, which does not derive from known genetic drivers.
Speed to Patient Impact
For patients and families living with ALS, time is not an abstraction. It is measured in months, sometimes weeks. Recent work targeting SOD1 mutations – though relevant to only a small percentage of patients – has delivered much-needed disease modifying therapeutics to patients. It has also moved the field forward by validating disease mechanisms, clinical endpoints, and development strategies that benefit the ALS community.
ALS carries an urgency that shapes every decision. Our approach with BMS reflects this urgency. By enabling collaborative yet differentiated programs – multiple targets, multiple modalities, pursued in parallel by insitro and BMS – we increase the odds that new therapies reach patients sooner. The imperative to help the many ALS patients who currently have no hope is a north star that shapes how we work, how we collaborate, and how we decide when to take bold swings – even, or perhaps especially, when tackling a problem that has proven to be so challenging.
An AI ALS disease axis → first-ever genetic screen for ALS reversion
About the insitro–Bristol Myers Squibb Collaboration
Launched in 2020, the collaboration combines insitro’s AI-enabled discovery platform with Bristol Myers Squibb’s neuroscience expertise to advance disease-modifying therapies for amyotrophic lateral sclerosis (ALS). Using insitro’s Virtual Human™ causal-biology engine, the
companies have identified multiple novel ALS targets (ALS-1, ALS-2, ALS-3) and are pursuing them across modalities. insitro will advance its own oligonucleotide program for ALS-1 while simultaneously progressing a small molecule for BMS. insitro’s TherML™ platform is guiding the design of the optimal intervention for each target.
